Laboratory professionals rely heavily on precision instruments to ensure accurate measurements and reliable results in their daily work. Among the most essential tools in any laboratory are disposable serological pipettes, which serve as the backbone for liquid handling procedures across various scientific disciplines. These graduated plastic instruments have revolutionized laboratory workflows by providing sterile, contamination-free solutions for transferring precise volumes of liquids. When selecting the right disposable serological pipettes for your laboratory needs, several critical factors demand careful consideration to ensure optimal performance, accuracy, and cost-effectiveness.
Understanding Volume Requirements and Accuracy Standards
Volume Range Selection
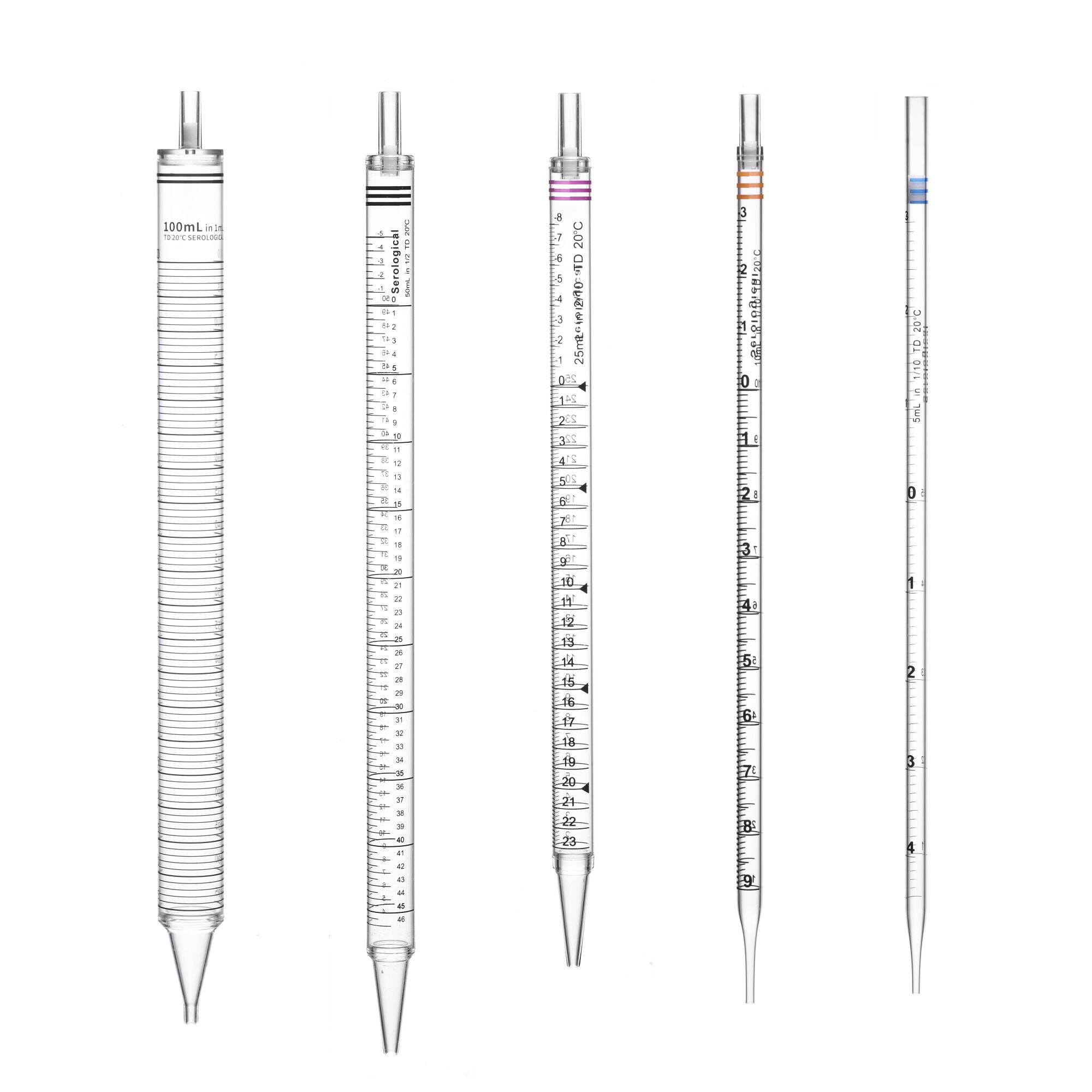
The selection of appropriate volume ranges represents one of the most fundamental considerations when purchasing disposable serological pipettes. Standard volumes typically range from 1ml to 100ml, with the most commonly used sizes being 5ml, 10ml, and 25ml pipettes. Your laboratory's specific applications will dictate which volumes you require most frequently. Cell culture work often demands 5ml and 10ml pipettes for media transfers, while larger volumes like 25ml and 50ml pipettes prove essential for buffer preparations and reagent handling.
Understanding your workflow requirements helps optimize inventory management and reduces unnecessary costs. Many laboratories benefit from maintaining a diverse range of volumes to accommodate various procedures, but careful analysis of usage patterns can reveal which sizes deserve priority in procurement decisions. Consider conducting a usage audit to identify the most frequently required volumes before making bulk purchases.
Accuracy and Precision Standards
Accuracy standards for disposable serological pipettes directly impact experimental reliability and reproducibility. High-quality pipettes should meet or exceed international standards such as ISO 8655 or ASTM specifications for volumetric accuracy. Typical accuracy tolerances range from ±2% for larger volumes to ±3% for smaller volumes, though premium products may offer tighter tolerances.
The graduation markings on pipettes must be clearly visible and precisely calibrated to ensure consistent measurements across different users and lighting conditions. Look for pipettes with bold, easy-to-read graduations that maintain their clarity throughout the product's shelf life. Some manufacturers offer color-coded graduation systems that enhance readability and reduce measurement errors in busy laboratory environments.
Material Quality and Manufacturing Standards
Plastic Composition and Purity
The plastic material used in manufacturing disposable serological pipettes significantly affects their performance and compatibility with various reagents. High-grade polystyrene remains the most common material choice due to its excellent clarity, chemical resistance, and moldability. Premium pipettes utilize virgin polystyrene that meets pharmaceutical-grade standards, ensuring minimal interference with sensitive biological samples or chemical reactions.
Material purity becomes particularly crucial when working with cell cultures, protein solutions, or other biological samples that can be adversely affected by plastic additives or impurities. Look for pipettes manufactured from materials that have been tested for endotoxin levels, DNA/RNA contamination, and cytotoxicity to ensure compatibility with your specific applications.
Manufacturing Process and Quality Control
The manufacturing process employed by different suppliers can vary significantly in terms of quality control measures and consistency standards. Reputable manufacturers implement comprehensive quality assurance programs that include statistical process control, batch testing, and traceability systems. These measures ensure that each pipette meets specified accuracy and sterility requirements consistently.
Injection molding techniques used in production should create smooth internal surfaces that minimize liquid retention and ensure complete drainage. Poor manufacturing can result in irregular internal surfaces that affect measurement accuracy and increase the risk of cross-contamination between samples. Quality manufacturers also implement rigorous cleaning procedures to remove any mold release agents or manufacturing residues that could interfere with laboratory applications.
Sterility and Contamination Prevention
Sterilization Methods and Validation
Sterility assurance represents a critical factor for laboratories working with cell cultures, microbiological samples, or other contamination-sensitive materials. Most high-quality disposable serological pipettes undergo gamma irradiation sterilization, which provides reliable sterility without leaving chemical residues or affecting plastic properties. This method ensures that pipettes remain sterile until their protective packaging is opened.
Validation of sterilization processes should be documented and traceable, with manufacturers providing certificates of sterility or compliance statements. Some applications may require specific sterility assurance levels, such as 10^-6 sterility assurance level for pharmaceutical applications. Understanding these requirements helps ensure that selected pipettes meet the necessary sterility standards for your specific laboratory needs.
Packaging and Individual Protection
Individual packaging systems play a crucial role in maintaining sterility and preventing contamination during storage and handling. Premium pipettes typically feature individual paper-plastic pouches or rigid containers that protect against physical damage while maintaining sterile barriers. The packaging material should be compatible with your laboratory's sterilization methods if additional sterilization is required.
Easy-open packaging designs improve workflow efficiency while minimizing contamination risks during pipette removal. Some packaging systems include color-coded labels or volume identification features that help prevent mix-ups in busy laboratory environments. Consider how packaging design will integrate with your laboratory's workflow and storage systems when evaluating different options.
Design Features and Ergonomic Considerations
Tip Design and Fluid Dynamics
The tip design of disposable serological pipettes significantly affects dispensing accuracy and user control during liquid handling procedures. Properly designed tips feature smooth, tapered ends that provide consistent flow rates while minimizing droplet formation and splashing. The tip opening should be appropriately sized for the intended volume range to ensure optimal flow characteristics.
Some advanced designs incorporate specialized tip geometries that reduce the formation of hanging drops, which can affect measurement accuracy and create potential contamination risks. Anti-drip tip designs help ensure that the last drop is delivered consistently, improving measurement precision and reducing sample waste in critical applications.
Grip Design and User Comfort
Extended laboratory sessions require ergonomic designs that minimize user fatigue and reduce the risk of repetitive strain injuries. Quality disposable serological pipettes feature textured gripping areas or specialized grip designs that provide secure handling even when wearing laboratory gloves. The overall length and weight distribution should feel balanced and comfortable during extended use.
Some manufacturers offer specialized grip designs with raised textures or finger positioning guides that enhance control and reduce slippage during precise manipulations. These features become particularly important when working with expensive reagents or irreplaceable samples where accuracy and control are paramount.
Compatibility and Application-Specific Requirements
Chemical Compatibility Assessment
Different laboratory applications involve various chemicals and solvents that may interact with pipette materials in unexpected ways. Polystyrene pipettes generally offer excellent compatibility with aqueous solutions, mild acids, and most biological buffers, but may not be suitable for organic solvents or strong acids. Understanding the chemical compatibility requirements for your specific applications helps prevent pipette degradation and potential contamination of samples.
Some specialized applications may require pipettes manufactured from alternative materials such as polypropylene or other chemically resistant plastics. These alternatives typically offer enhanced chemical resistance but may come at a higher cost or with different performance characteristics. Evaluating the full range of chemicals and solutions used in your laboratory ensures appropriate material selection.
Temperature and Storage Considerations
Operating temperature ranges can affect both pipette performance and material integrity over time. Standard polystyrene pipettes typically perform well at room temperature but may become brittle at very low temperatures or soften at elevated temperatures. Applications involving temperature-sensitive procedures may require pipettes with enhanced temperature stability or specific storage requirements.
Long-term storage conditions also influence pipette quality and performance. Exposure to direct sunlight, extreme temperatures, or high humidity can degrade plastic materials and affect measurement accuracy. Proper storage in controlled environments helps maintain pipette quality and extends shelf life, ultimately providing better value for your investment.
Cost Analysis and Value Optimization
Total Cost of Ownership Evaluation
While unit cost often drives purchasing decisions, a comprehensive total cost of ownership analysis provides better insight into long-term value. This analysis should include factors such as accuracy-related rework costs, contamination risks, user productivity, and inventory management expenses. Higher-quality pipettes may justify their premium pricing through reduced error rates and improved laboratory efficiency.
Bulk purchasing arrangements can provide significant cost savings, but inventory carrying costs and shelf life considerations must be balanced against volume discounts. Many suppliers offer flexible purchasing programs that allow laboratories to optimize their cost structure while maintaining adequate inventory levels for operational needs.
Supplier Reliability and Support Services
Supplier reliability affects both cost predictability and operational continuity in laboratory settings. Established suppliers with strong distribution networks can provide consistent product availability and technical support when issues arise. Evaluate suppliers based on their quality certifications, regulatory compliance history, and customer service capabilities.
Technical support services, including application assistance and troubleshooting guidance, add significant value beyond the basic product cost. Suppliers who provide comprehensive documentation, training resources, and responsive customer service help ensure optimal product performance and user satisfaction throughout the product lifecycle.
FAQ
What volume accuracy can I expect from quality disposable serological pipettes
Quality disposable serological pipettes typically offer accuracy within ±2-3% of the stated volume, with premium products achieving even tighter tolerances. The accuracy depends on proper manufacturing standards, calibration procedures, and adherence to international specifications such as ISO 8655. Always verify that pipettes meet the accuracy requirements for your specific applications and consider conducting periodic validation checks to ensure continued performance.
How long can sterile disposable serological pipettes be stored before use
Most sterile disposable serological pipettes maintain their sterility for 3-5 years when stored properly in unopened original packaging. Storage should occur in clean, dry conditions away from direct sunlight and extreme temperatures. Always check expiration dates and manufacturer recommendations, as shelf life can vary depending on packaging materials and sterilization methods used.
Are there specific considerations for cell culture applications
Cell culture applications require pipettes that are not only sterile but also free from endotoxins, DNA/RNA contamination, and cytotoxic substances. Look for pipettes specifically certified for cell culture use, manufactured from pharmaceutical-grade materials, and tested for biological compatibility. Individual packaging helps maintain sterility, while smooth internal surfaces minimize cell damage during transfers.
What should I do if pipettes show inconsistent measurements
Inconsistent measurements may indicate manufacturing defects, improper storage, or user technique issues. First, verify proper pipetting technique and ensure pipettes are being used within their specified temperature and chemical compatibility ranges. If problems persist, contact your supplier for technical support and consider switching to a different lot or manufacturer. Document any accuracy issues for quality control purposes and potential warranty claims.




